Ultra‑Early Cancer Detection With cfDNA Blood Tests: China vs USA in the New Oncology Race
When standard scans show nothing but cancer is silently growing, ultra‑early detection can be the difference between a curable disease and a late‑stage emergency. Circulating cell‑free DNA (cfDNA) blood tests are emerging as one of the most exciting tools in this new era of oncology, with China and the United States pushing the science forward in different ways. [ncbi.nlm.nih]
For patients and doctors exploring international treatment options, understanding what cfDNA can and cannot do is now a practical question – not just a scientific curiosity. [pmc.ncbi.nlm.nih]
Figure: cfDNA analysis process showing methylation patterns and fragmentomics
What Is cfDNA and Why Does It Matter?
All cells in the body, healthy or cancerous, release tiny fragments of DNA into the bloodstream when they die. This fragmented genetic material is called cell‑free DNA (cfDNA). [nature]
When the fragments originate from tumor cells, they are called circulating tumor DNA (ctDNA) and carry cancer‑specific genetic and epigenetic signals. [pmc.ncbi.nlm.nih]
Modern sequencing and AI‑based analysis can interrogate cfDNA for:
- Somatic mutations (cancer‑driving genetic changes). [pmc.ncbi.nlm.nih]
- Methylation patterns and other epigenetic markers. [nature]
- Fragmentomics – the length, end‑motifs and distribution of cfDNA fragments, which differ between healthy and cancer states. [pubmed.ncbi.nlm.nih]
The clinical promise is simple to understand but technically complex:
- A single blood draw could one day screen for multiple cancers at once.
- The same technology can monitor minimal residual disease (MRD), relapse and response to therapies like chemotherapy, targeted agents and cell therapies. [pmc.ncbi.nlm.nih]
From Idea to Clinic: A Short History of cfDNA in Cancer
The evolution of cfDNA in oncology can be summarized in four main phases:
1. Discovery Era
Early studies showed that cancer patients had higher levels of fragmented DNA in their blood than healthy individuals, suggesting that a "liquid biopsy" might be possible. [pmc.ncbi.nlm.nih]
These first reports were proof‑of‑concept and focused largely on total cfDNA quantity and simple mutation detection.
2. Liquid Biopsy for Advanced Disease
With the advent of next‑generation sequencing (NGS), cfDNA became a powerful tool to:
- Detect actionable mutations in advanced lung, colorectal and breast cancers.
- Follow clonal evolution and emerging resistance mutations under targeted therapies. [pmc.ncbi.nlm.nih]
This stage firmly established cfDNA as a non‑invasive complement to tissue biopsy in advanced cancer management.
3. Early Detection and Multi‑Cancer Tests
The next step was much more ambitious: using cfDNA to detect cancer before symptoms or visible lesions appear on imaging. [pmc.ncbi.nlm.nih]
Chinese cohorts pioneered studies where stored blood samples from apparently healthy individuals were later re‑analyzed once some of them developed cancer, showing that cancer‑associated signatures could be detected several years prior to clinical diagnosis. [pmc.ncbi.nlm.nih]
In parallel, US‑based multi‑cancer early detection (MCED) programs began to explore methylation, fragmentomics and machine learning to detect dozens of cancer types and predict tissue of origin from a single sample. [nature]
4. Fragmentomics and Deep Profiling
Recent work has shown that not only "what" is in cfDNA matters, but "how" it is cut and arranged in the genome. [ncbi.nlm.nih]
Deep profiling of cfDNA fragment ends and motifs can distinguish early‑stage cancers (including colorectal and renal cancers) from healthy controls with high accuracy, and can be integrated with mutation and methylation analysis. [ncbi.nlm.nih]
What Can Current cfDNA Tests Actually Do?
Despite the headlines, most patients and doctors need clear, practical categories rather than hype. Today, cfDNA is used or actively evaluated in four main scenarios:
1. Minimal Residual Disease (MRD) and Relapse Monitoring
- Detecting microscopic residual disease after surgery or systemic therapy in colorectal, breast and hematologic cancers. [pmc.ncbi.nlm.nih]
- Identifying molecular relapse months before it becomes visible on imaging or causes symptoms. [pmc.ncbi.nlm.nih]
This has direct implications for:
- Tailoring adjuvant therapy (escalation or de‑escalation).
- Designing more personalized surveillance schedules.
2. Therapy Response Evaluation
Serial cfDNA measurements can show:
- Rapid decline in ctDNA levels in responders.
- Plateau or re‑emergence of ctDNA in non‑responders or those developing resistance. [pmc.ncbi.nlm.nih]
For advanced therapies such as CAR‑T, TIL or NK cell therapy, cfDNA‑based MRD may soon become a key component of response assessment.
3. Single‑Cancer Early Detection and High‑Risk Screening
Several studies have evaluated cfDNA‑based detection for specific cancers such as lung, colorectal and liver cancer, especially in high‑risk populations. [pmc.ncbi.nlm.nih]
These tests focus on:
- Known driver mutations or methylation signatures.
- Tumor‑informed assays tailored to each patient's tumor profile.
4. Multi‑Cancer Early Detection (MCED)
Multi‑cancer platforms aim to:
- Detect a cancer signal.
- Predict tissue of origin (e.g., lung, liver, colon, pancreas). [nature]
Randomized studies are now exploring MCED in individuals with hereditary cancer syndromes and elevated risk, to evaluate clinical effectiveness and downstream impact on outcomes. [nature]
Comparing the Leaders: China's cfDNA Tech vs. GRAIL's Galleri Test
China's cfDNA technologies and GRAIL's Galleri test represent two leading but distinct approaches to multi-cancer early detection (MCED) using cell-free DNA analysis. Both aim to spot cancer signals years before symptoms through a simple blood draw, but they differ in methodology, development stage, regulatory status, and practical availability.
| Aspect | China's cfDNA Tech (e.g., PanSeer by Singlera Genomics) | GRAIL Galleri Test |
|---|---|---|
| Primary Biomarker | cfDNA methylation patterns, often combined with fragmentomics [pmc.ncbi.nlm.nih] | Targeted methylation sequencing of cfDNA, powered by machine learning classifiers [grail] |
| Cancers Detected | Primarily 5 types (stomach, esophageal, colorectal, lung, liver); potential for expansion [pmc.ncbi.nlm.nih] | 50+ cancer types, including hard-to-detect ones like pancreatic and ovarian [grail] |
| Early Detection Window | Up to 4 years pre-diagnosis in key studies (e.g., PanSeer on 123,000+ Chinese cohort) [pmc.ncbi.nlm.nih] | Stage I-III sensitivity ~40-50% at 99.5% specificity; real-world PPV ~49% [pmc.ncbi.nlm.nih] |
| Tissue of Origin (CSO) | Limited prediction; focuses more on signal detection [nature] | High accuracy (>90%) in predicting cancer origin via methylation classifiers [grail] |
| Key Strength | Large population studies in high-risk groups; retrospective proof-of-concept [pmc.ncbi.nlm.nih] | Broad coverage, validated in diverse cohorts; strong CSO for clinical follow-up [grail] |
Important Clarification: The "8-Year Early Detection" Claim
No specific Chinese clinical trial or validated method exists for cfDNA-based cancer detection precisely 8 years before symptoms. The "8 years" claim appears to be an exaggeration. The primary source is the PanSeer study, a retrospective analysis that showed a detection window of up to 4 years pre-clinical diagnosis for 5 cancers, not a prospective clinical trial. [pmc.ncbi.nlm.nih]
Limitations and Ethical Challenges: Beyond the Hype
An article that is truly useful for patients and doctors must state the limitations clearly:
- Low ctDNA fraction in early disease: In very early cancer, ctDNA may account for less than 0.01% of total cfDNA, making detection extremely challenging and potentially leading to false negatives. [nature]
- False positives and overdiagnosis: cfDNA tests may detect biologically indolent lesions that would never have caused symptoms, creating anxiety and leading to unnecessary interventions. [pmc.ncbi.nlm.nih]
- Uncertain tissue of origin or clinical pathway: A "cancer signal detected" result without a clearly localized lesion can trigger complex, invasive and costly diagnostic cascades. [pmc.ncbi.nlm.nih]
- Equity and access: Advanced cfDNA tests are expensive and currently concentrated in major academic centers and specialized programs, raising questions about global accessibility. [nature]
Ethically, responsible use demands:
- Robust informed consent processes explaining benefits, risks and uncertainties.
- Clear algorithms for follow‑up imaging and biopsies.
- Strong data‑privacy protections for genomic information.
China vs USA: Two Different, Complementary Strengths
For a platform like CancerCareE coordinating advanced treatments across continents, understanding the differences between Chinese and US ecosystems is critical.
China: High Volume, Fast Innovation
China has rapidly become a global leader in both cell therapies (CAR‑T, NK, TIL, Gamma Delta T‑cells) and cfDNA‑based liquid biopsy research. [pmc.ncbi.nlm.nih]
Key characteristics include:
- Very large patient cohorts and high volumes of hematologic and solid tumor cases.
- Hundreds of registered CAR‑T and cell therapy trials, often integrated with genomic and cfDNA‑based monitoring.
- Rapid integration of new assays into clinical research settings, especially in major hubs such as Beijing, Shanghai and Hainan's Boao Lecheng International Medical Tourism Pilot Zone.
China's Practical Advantages:
- Speed and scale with huge cohorts; ideal for Asia-prevalent cancers.
- Often bundled with advanced therapies (e.g., cell therapies).
- Potentially lower cost in clinical trial settings.
China's Current Limitations:
- MCED tests are primarily research-based, not approved for population screening.
- Narrower cancer coverage compared to platforms like Galleri.
- Less emphasis on precise tissue of origin prediction.
USA: Stringent Regulation and Guideline‑Driven Adoption
The US system is characterized by:
- Strong regulatory oversight by the FDA and payers, requiring robust evidence before widespread adoption of new tests. [pmc.ncbi.nlm.nih]
- Emphasis on randomized trials and health‑economics analyses to demonstrate that early detection improves survival and is cost‑effective. [nature]
- Integration of cfDNA into practice guidelines mainly in clearly defined contexts (e.g., MRD in certain cancers, targeted therapy selection).
Galleri's Practical Advantages:
- Broad cancer coverage (50+ types) with high accuracy in predicting tissue of origin.
- Rigorous prospective validation reduces overdiagnosis risk.
- Commercially available with a clear clinical pathway.
Galleri's Current Limitations:
- Higher cost (~$949).
- Sensitivity drops for very early stages.
- Primarily US-centric access.
Which Is "Better" for a Patient?
There is no universal answer. The right choice depends on:
- Disease stage and biology.
- Previous treatments and remaining standard options.
- The patient's risk tolerance, financial constraints and willingness to travel for trials.
For CancerCareE users seeking advanced options:
- Consider China for trial access combining cfDNA monitoring with CAR-T/TIL/NK in high-volume centers—ideal for relapsed cases seeking integrated, cutting-edge protocols.
- Consider Galleri/US for broad, validated screening with a clear diagnostic pathway, coordinated via US partners for integrated care.
- A Hybrid Approach may be optimal: using Galleri for initial signal detection, then leveraging China's advanced therapy and trial infrastructure for treatment.
Practical Value for Patients and Physicians
For Patients
- cfDNA tests can provide additional information, but they are not a magic crystal ball. They should be interpreted by experienced oncologists within a full clinical context.
- Patients exploring international treatment (China, USA, Europe, Asia) should ask specifically how cfDNA and liquid biopsy are used for:
- Eligibility assessment for advanced therapies.
- Monitoring response and relapse.
- Clinical decision‑making beyond imaging and standard labs.
For Physicians
- cfDNA is best viewed as a complementary biomarker, not a replacement for histopathology and imaging.
- When referring patients abroad for CAR‑T, NK, TIL or other advanced modalities, clarifying how partner centers use cfDNA and MRD can help align expectations and follow‑up plans.
- Collaboration with medical tourism coordinators who understand both the scientific and logistical dimensions can streamline patient journeys.
Ready to Explore Advanced Cancer Treatment Options?
If you're considering international cancer treatment and want to understand how cfDNA monitoring and advanced therapies like CAR‑T could be integrated into your treatment plan, our medical coordination team can help.
Get Free Preliminary AssessmentBottom Line for Patients
China leads in volume and integration of cfDNA with advanced cell therapies within clinical trials, offering fast, cost-effective access via medical tourism. The US, through platforms like GRAIL's Galleri, leads in validated, broad-spectrum MCED with strong regulatory oversight. The "8-year early detection" claim is not yet a clinical reality.
By prioritizing transparency, scientific rigor and patient‑centered communication, CancerCareE can turn the global "cfDNA race" between China and the USA into a real, practical advantage for patients who need options beyond standard care.
About CancerCareE
We coordinate access to advanced cancer therapies including CAR‑T, Gamma Delta T‑cells, TIL and NK therapies through partner hospitals across Europe, USA, and Asia.
Related Articles
Multilingual Versions
Related Articles
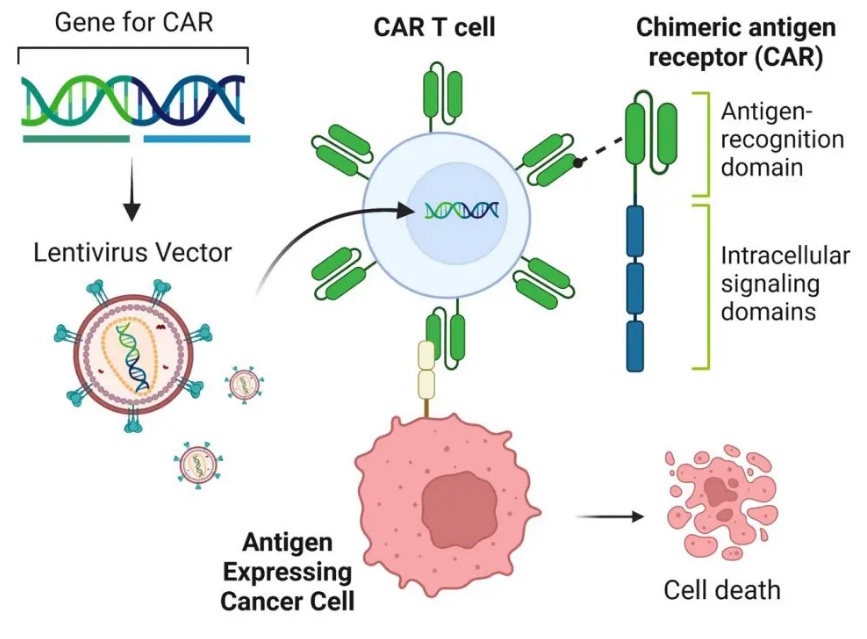
CAR‑T Therapy: Complete Guide for Patients
Everything you need to know about CAR‑T therapy, including eligibility, process, costs, and international access options.
Read More
Medical Tourism to China: Complete Guide
Practical information for international patients considering cancer treatment in China's top hospitals.
Read More
Immunotherapy Advances in 2026
The latest developments in cancer immunotherapy, including checkpoint inhibitors and combination therapies.
Read More