Photo-Immunotherapy: The Japanese Light-Based Surgery Transforming Cancer Treatment
For Patients and Families: Your Guide to Photo-Immunotherapy
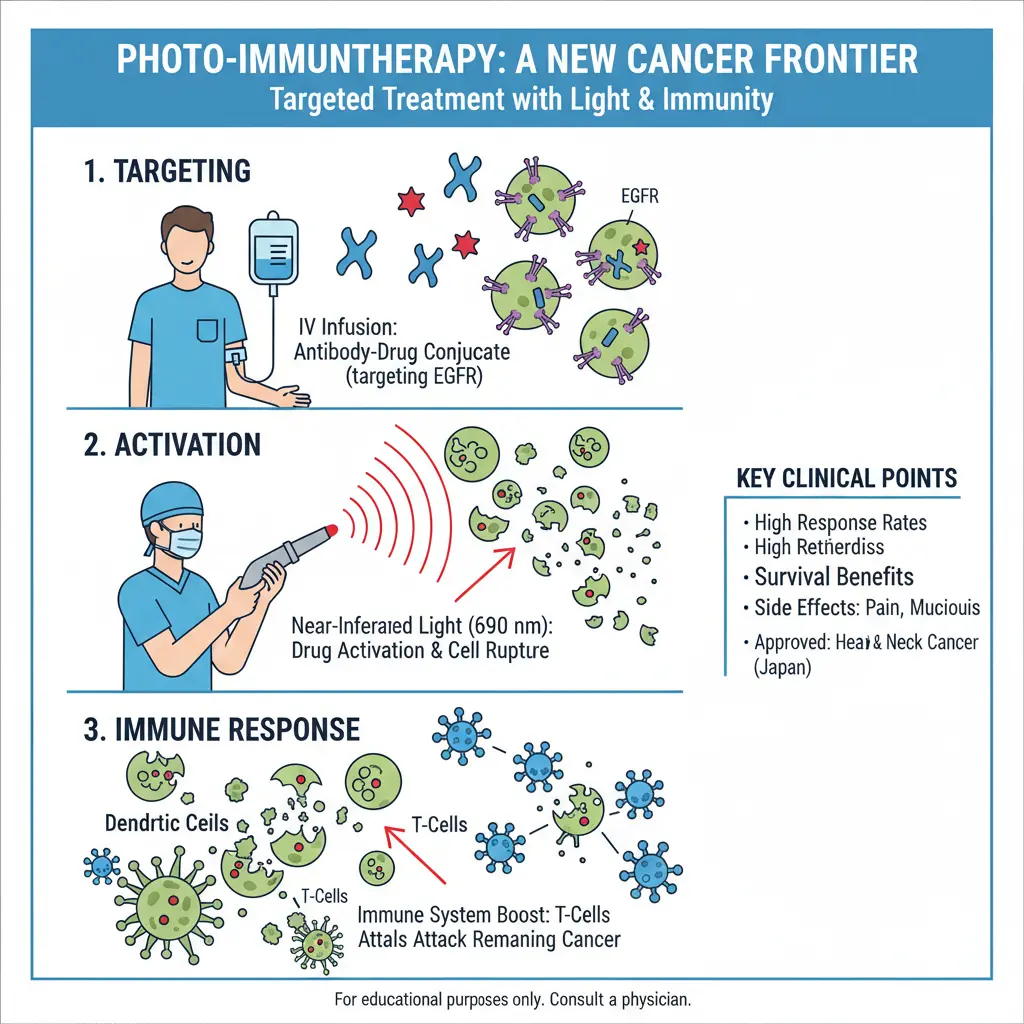
What Is Photo-Immunotherapy in Simple Terms?
Imagine a treatment that works like this:
- Step 1: You receive an intravenous infusion of a "smart" drug that seeks out and attaches only to your cancer cells—like a GPS tracker locked onto a specific target.
- Step 2: About a day later, a surgeon shines a special red laser light directly onto your tumor.
- Step 3: The light activates the drug, causing the cancer cells to swell up and burst—literally within minutes. And here's the remarkable part: as those cancer cells die, they send out signals that wake up your immune system to attack any remaining cancer cells throughout your body.
This isn't science fiction. It's near-infrared photoimmunotherapy (NIR-PIT), and since 2021, it has been available in Japan for patients with unresectable head and neck cancer.
Who Is This Treatment For?
Photo-immunotherapy is currently approved in Japan for patients with:
- Locally advanced or recurrent head and neck cancer that cannot be removed surgically
- Tumors that express EGFR (a protein on the surface of cancer cells)—testing is required before treatment
- Patients who have already tried standard treatments like chemotherapy, radiation, or both
It is typically NOT suitable for:
- Patients with widespread distant metastases (though research is exploring this)
- Those who cannot safely undergo general anesthesia
- Patients whose tumors show low or no EGFR expression
- People without access to a specialized treatment center equipped with the laser system
What Actually Happens on Treatment Day?
Day 1 (Infusion Day): You arrive at the hospital and receive the antibody-drug conjugate (cetuximab sarotalocan, brand name Akalux®) through an IV. The infusion itself takes about an hour. You then go home or stay in the hospital while the drug circulates and binds to your cancer cells over the next 20-28 hours.
Day 2 (Treatment Day): You're taken to an operating room and placed under general anesthesia. The surgeon positions a laser fiber to deliver red light (690 nm wavelength) precisely to the tumor. The light exposure typically lasts minutes to hours depending on the tumor size and location. Afterward, you're monitored closely as you wake up.
The Days and Weeks After: Most patients experience some pain and swelling in the treated area—this is expected and managed with medications. You may need temporary feeding support if the tumor is in the mouth or throat. Your medical team will schedule follow-up imaging (usually CT or PET scans) about 4-8 weeks later to assess the response.
Common Questions Patients Ask
Does it hurt?
During the procedure, you're under anesthesia, so you feel nothing. Afterward, pain at the treatment site is very common—reported in over 90% of patients—but it's typically managed effectively with standard pain medications.
Will I have a scar?
Unlike traditional surgery, there's no surgical incision. However, if the tumor was near the skin surface, there may be some local skin changes. Internally, the treated area will heal like any other wound.
How many treatments can I have?
Some patients receive multiple sessions. The decision depends on your response, overall health, and whether new lesions appear in accessible locations.
Can this help if my cancer has spread to other parts of my body?
The treatment itself is local—it only affects areas exposed to light. However, because it triggers an immune response, there is potential for "abscopal effects" where the immune system attacks distant tumors. This is an area of active research, and results vary by patient.
For Clinicians: A Practical Overview
Current Indication
Near-infrared photoimmunotherapy (NIR-PIT) with cetuximab sarotalocan (Akalux®) is approved in Japan for patients with unresectable, locally advanced or recurrent head and neck squamous cell carcinoma (HNSCC) that expresses EGFR, after failure of standard therapies.
Key Clinical Data (Real-World Outcomes)
Procedure Workflow
- Patient selection: Confirm unresectable status, EGFR positivity (by IHC), adequate performance status, and airway safety.
- Infusion: Administer cetuximab sarotalocan IV (standard dose: 640 mg/m²).
- Light activation: 20-28 hours post-infusion, under general anesthesia, deliver 690 nm laser light at 50-150 J/cm² (dose depends on tumor depth and location).
- Post-procedure monitoring: Observe for pain, edema, airway compromise, bleeding, and fistula formation. Multidisciplinary support (pain management, nutrition, airway protection) is often required.
Adverse Events (Real-World Data)
| Adverse Event | Incidence | Grade ≥3 |
|---|---|---|
| Pain | 92.5% | 17.5% |
| Mucositis/stomatitis | 80.0% | 17% |
| Laryngeal edema | ~67% | 8% |
| Dysphagia | ~50% | Data not specified |
| Bleeding | ~50% | Data not specified |
| Fistula | ~33% | Data not specified |
Proactive airway management (including preventive tracheostomy in selected cases) and temporary enteral nutrition are frequently necessary.
Patient Selection Considerations
- Strongest predictor of response: EGFR expression level (higher expression correlates with better outcomes)
- Contraindications: Inability to tolerate general anesthesia, severe uncorrectable coagulopathy, tumor invasion into major blood vessels where light delivery is impossible
- Relative considerations: Prior radiation to the same site is NOT a contraindication; many treated patients have had previous radiotherapy
The Science Made Clear: How Photo-Immunotherapy Works
The Mechanism: A Double Punch
Punch 1: Physical Destruction
The drug (cetuximab-IR700) binds to EGFR on cancer cells. When exposed to 690 nm near-infrared light, the dye undergoes a structural change that makes it hydrophobic. This causes the antibody-dye complexes to aggregate and physically distort the cell membrane—leading to rapid swelling and rupture. This happens within minutes, independent of oxygen, making it effective even in hypoxic tumor environments.
Punch 2: Immunogenic Cell Death (ICD)
As cancer cells burst, they release damage-associated molecular patterns (DAMPs) and tumor antigens. This "danger signal" recruits dendritic cells, which present the antigens to T-cells, effectively turning the tumor into an in-situ vaccine. The result is a systemic immune response that can potentially attack metastases not exposed to light.
What the Latest Research Shows (2025-2026)
Beyond Head and Neck Cancer
While currently approved only for HNSCC, research is rapidly expanding:
| Cancer Type | Target | Status |
|---|---|---|
| Breast cancer (TNBC) | ICAM-1 | Preclinical—effective in mouse models |
| Bladder cancer (NMIBC) | EGFR, Nectin-4, TROP-2 | Preclinical—potent cell killing |
| Pancreatic cancer | Stromal targeting | Preclinical—remodels TME |
| Ovarian cancer | EGFR, Her2, FOLR1, TROP-2, TF | Preclinical—ICD induction confirmed |
| Neuroblastoma (pediatric) | GD2 | Preclinical—significant tumor suppression |
Combination Strategies in Development
- CD25-targeted PIT + IL-15 + anti-PD-1: Depletes regulatory T-cells in the tumor microenvironment, enhancing CD8+ T-cell activity. In mouse models, this combination achieved 90% complete responses.
- FAP-targeted PIT for drug delivery: By destroying cancer-associated fibroblasts, this approach reduces the physical barrier to chemotherapy penetration.
- STING agonist combinations: Activating the innate immune pathway synergizes with PIT-induced ICD for stronger antitumor immunity.
Limitations and Unanswered Questions
- Most data come from a single country (Japan) with a specific healthcare system
- Patient numbers in published studies remain relatively small (tens to low hundreds)
- Long-term outcomes beyond 2-3 years are still being collected
- The treatment requires specialized equipment and multidisciplinary teams, limiting accessibility
- Not all patients respond—EGFR expression level is critical, and even some EGFR-positive tumors show resistance
- The optimal number of treatment sessions and retreatment intervals are still being defined
How to Talk With Your Doctor About Photo-Immunotherapy
If you or a loved one has advanced head and neck cancer and standard treatments are no longer working, here are questions to guide a productive conversation with your oncologist:
- "Has my tumor been tested for EGFR expression? If not, can we test it?" (This is essential—the treatment only works if your cancer has enough EGFR.)
- "Based on my situation—tumor location, prior treatments, and overall health—do you think I might be a candidate for photo-immunotherapy?"
- "Do you have any experience with this treatment, or do you know colleagues or centers that do?" (Even if your doctor can't offer it, they may know referral pathways or clinical trials.)
- "Are there any clinical trials in the United States, Europe, or Japan that I might qualify for?"
- "If I were to consider traveling to Japan for this treatment, what medical records and tests would I need to prepare?"
How CancerCareE Can Help
At CancerCareE, we specialize in coordinating access to advanced cancer treatments worldwide. While photo-immunotherapy is currently only approved in Japan, we can:
- Help you understand if you might be a candidate based on your medical records
- Connect you with partner hospitals in Japan that perform this procedure
- Coordinate the entire medical travel process including visa assistance, hospital appointments, translation services, and accommodation
- Provide ongoing support throughout your treatment journey
Important note: We do not provide medical treatment ourselves. We coordinate access to accredited hospitals and medical providers. All medical decisions are made by licensed physicians at partner institutions.
Ready to Explore Your Options?
Submit your medical records for a preliminary review by our coordination team. We'll help you understand if photo-immunotherapy or other advanced treatments might be appropriate for your case.
The Bottom Line: Hope With Honesty
Photo-immunotherapy represents a genuine advance in the treatment of unresectable head and neck cancer. For carefully selected patients, it offers:
- High response rates (75% in real-world data)
- Meaningful survival extension (median OS 26.9 months)
- Precision targeting with minimal damage to surrounding healthy tissue
- Immune activation that may provide additional long-term benefit
But it is not a miracle cure for everyone. It requires specialized centers, expert multidisciplinary teams, and careful patient selection. The data, while impressive, come from relatively small cohorts and need validation in larger, international trials.
For the right patient, however, this "light-based surgery from Japan" is quite literally a new ray of hope.
Disclaimer: This information is for educational purposes and is not a substitute for professional medical advice. Always consult with a qualified healthcare provider for diagnosis and treatment options. Photo-immunotherapy with cetuximab sarotalocan is approved in Japan but may not be available in other countries. Clinical trials may be an option for patients outside Japan.
References
- Tahara H, et al. Outcomes of Near-Infrared Photoimmunotherapy for Head and Neck Cancer: A Single-Center Retrospective Study. Cancers. 2026;18(3):350.
- Dynarowicz K, et al. A Decade of Innovation in Breast Cancer (2015-2025): A Comprehensive Review. Cancers. 2026;18(3):361.
- Suzuki S, et al. Predictive Value of EGFR Expression for the Efficacy of Near-Infrared Photoimmunotherapy in Head and Neck Cancer. JMA Journal. 2026.
- Hirakawa H, et al. Preliminary observations on peripheral inflammatory markers in NIR-PIT and ICI-treated recurrent head and neck squamous cell carcinoma. Eur Arch Otorhinolaryngol. 2026.
- Mohiuddin TM, et al. Synthetic zipper mediated pre-targeting system for near-infrared photoimmunotherapy. iScience. 2026.
- Nouso H, et al. Near-infrared Photoimmunotherapy Targeting High-risk Human Neuroblastoma Cells Expressing GD2. Anticancer Res. 2025.